Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV
Por um escritor misterioso
Descrição

Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV-2: A preliminary report of an open-label, Phase 1 clinical trial - eClinicalMedicine

Intradermal DNA vaccine delivery using vacuum-controlled, needle-free electroporation: Molecular Therapy - Nucleic Acids

Positive Results from Preclinical Testing Support Clinical Development of COVID-19 DNA Vaccine
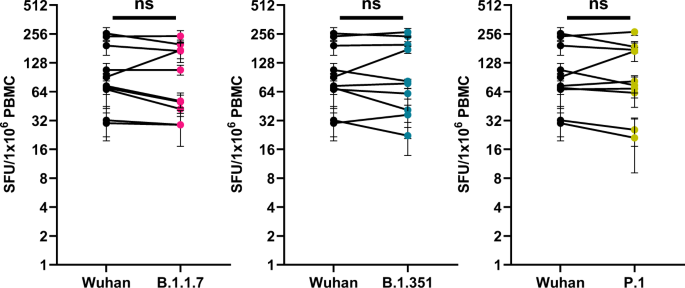
INO-4800 DNA vaccine induces neutralizing antibodies and T cell activity against global SARS-CoV-2 variants
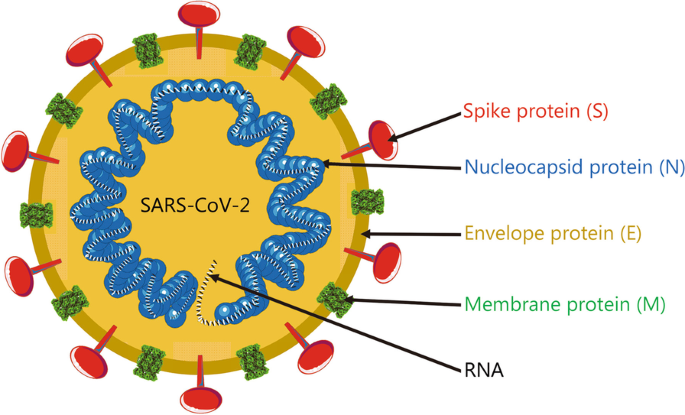
Advances in the design and development of SARS-CoV-2 vaccines, Military Medical Research

Phase 1 U.S. Trial of COVID-19 DNA Vaccine Enrollment Complete
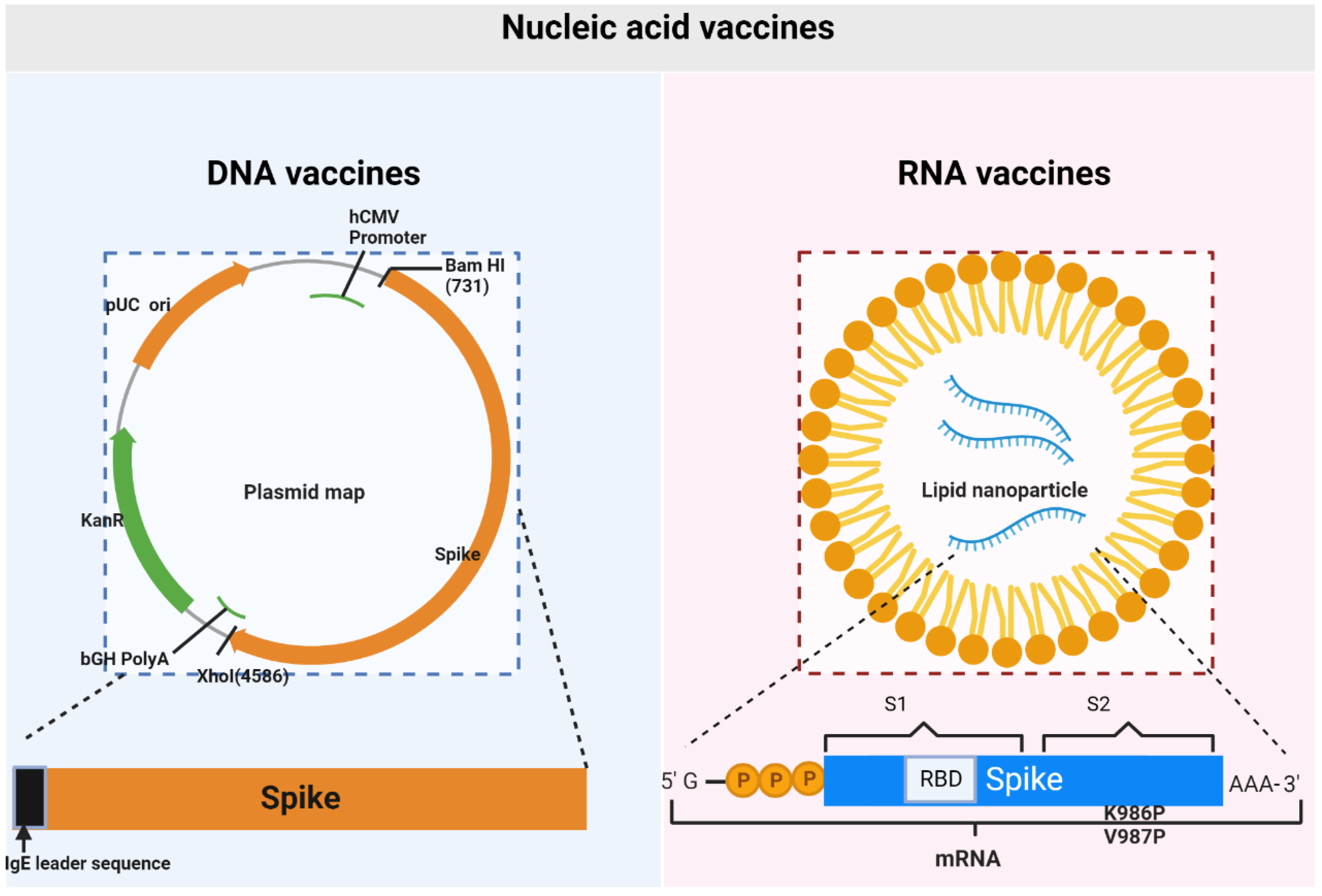
Vaccines, Free Full-Text

COVID-eVax, an electroporated DNA vaccine candidate encoding the SARS-CoV-2 RBD, elicits protective responses in animal models - ScienceDirect
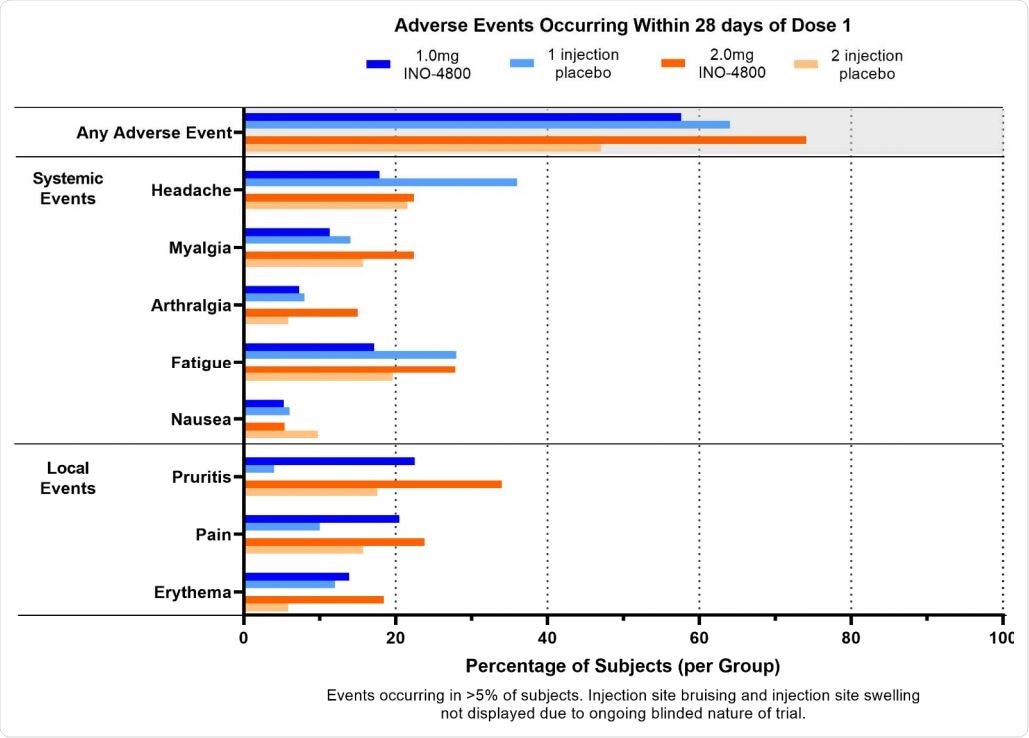
Phase 2 trial shows INO-4800 SARS-CoV-2 DNA vaccine safe and tolerable in adults

Innovative Vaccines to Fight COVID-19, Other Viruses - IEEE Pulse
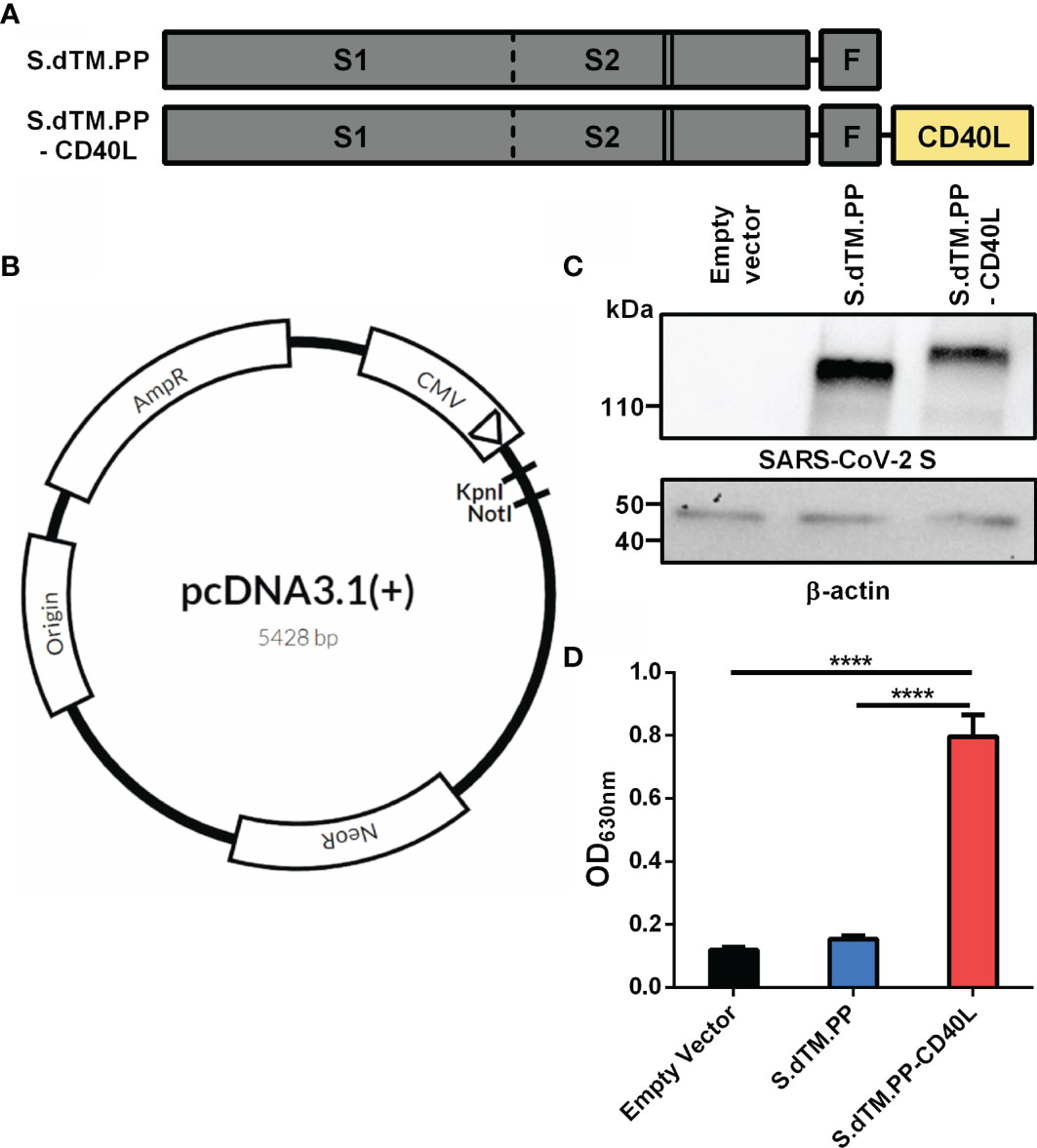
Frontiers DNA Based Vaccine Expressing SARS-CoV-2 Spike-CD40L Fusion Protein Confers Protection Against Challenge in a Syrian Hamster Model

Safety, tolerability, and immunogenicity of a SARS-CoV-2 recombinant spike RBD protein vaccine: A randomised, double-blind, placebo-controlled, phase 1-2 clinical trial (ABDALA Study) - eClinicalMedicine

A Comprehensive Review of the Global Efforts on COVID-19 Vaccine Development
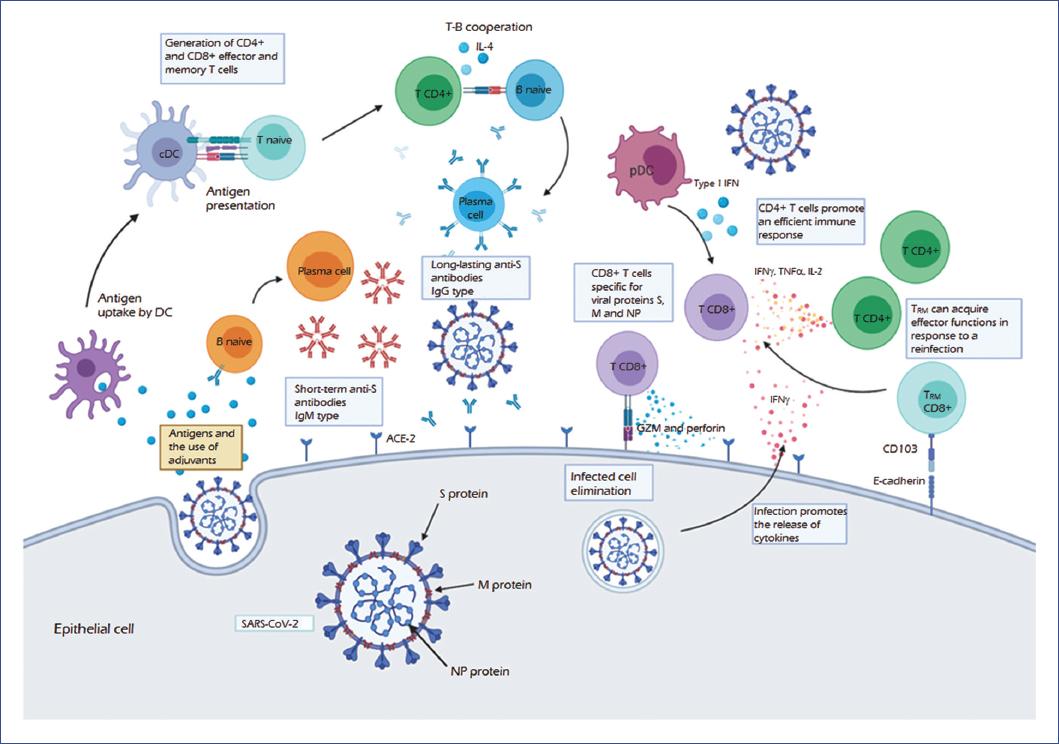
SARS-CoV-2: previous coronaviruses, immune response, and development of vaccines
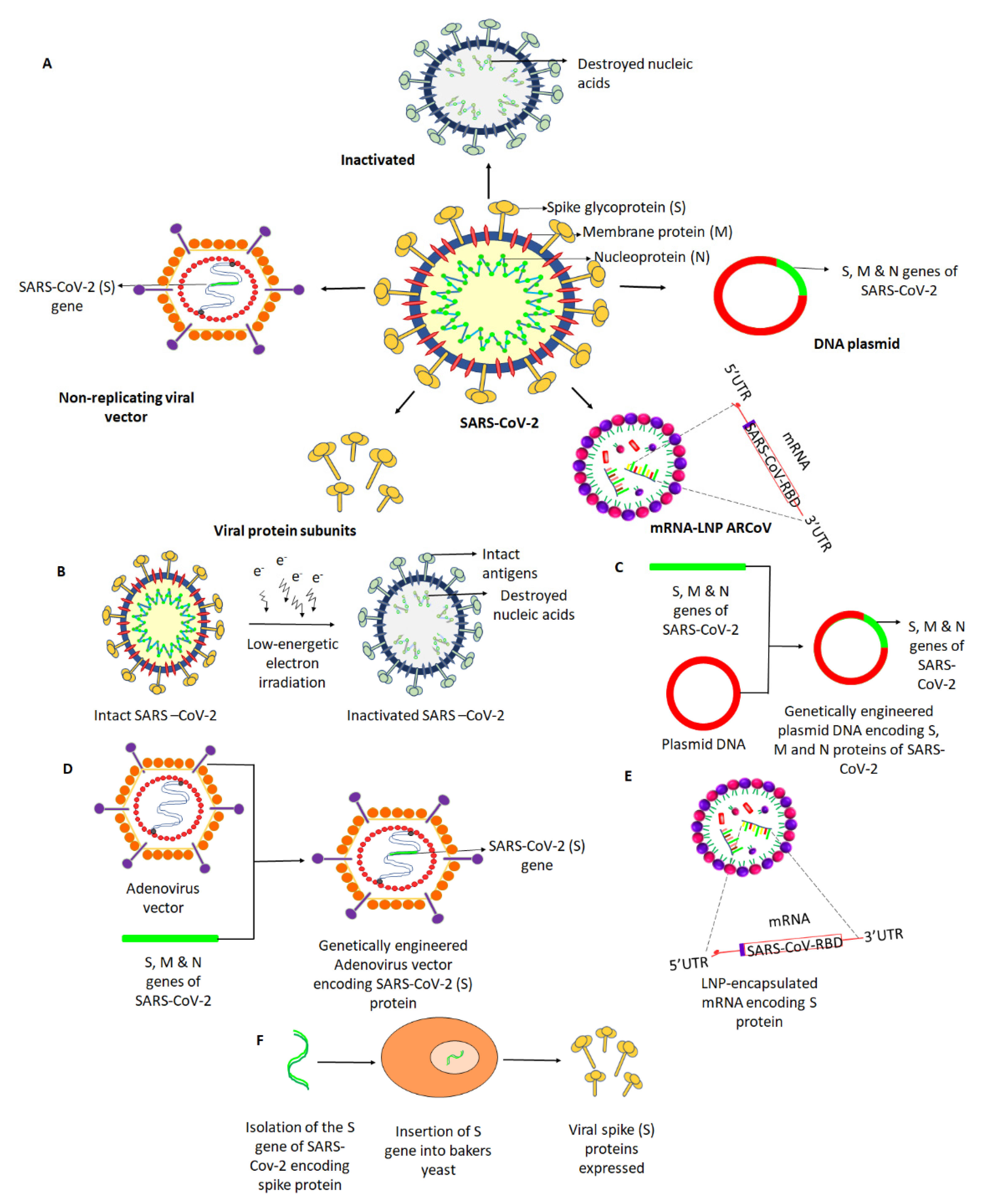
Vaccines, Free Full-Text
de
por adulto (o preço varia de acordo com o tamanho do grupo)