CSPI asks FDA to ban powdered caffeine sold as a dietary supplement
Por um escritor misterioso
Descrição
The Center for Science in the Public Interest (CSPI) has sent a petition to federal regulators seeking the ban of pure, powdered caffeine that is packaged and sold as a dietary supplement. Because of the product’s extreme potency, the possibility of accidental overdose poses a clear and present public health risk, the organization asserts.

FDA cracks down on illegal sales of pure caffeine
Buying Pure Caffeine Powder Off the Internet

PDF) Policy Recommendations to Address Energy Drink Marketing and Consumption by Vulnerable Populations in the United States

FDA Advisory No. 2019-536

Wellspring Issue #30 by The Wellspring - Issuu
Appendix A: Legal Aspects of the Food Additive Approval Process, Enhancing the Regulatory Decision-Making Approval Process for Direct Food Ingredient Technologies

FDA Warns Against Concentrated Caffeine (Pure Powder & Liquids)

Nutrition Page 2There

Kidney Stones In Kindergarten? Put Down the Sports Drink, Kiddo!

Posts by Ari Goldstein, Ph.D. — Educational Psychologist
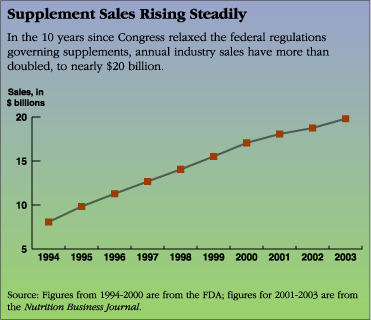
CQ Researcher - Dietary Supplements
FDA Going After Sellers of Pure Caffeine Powder
CSPI asks FDA for high potency caffeine ban
de
por adulto (o preço varia de acordo com o tamanho do grupo)







